Homepage
300,000 EU citizens call on German environmental minister Steffi Lemke: Stop Gene Drives!
Berlin, 31 May 2022 – Almost 300,000 citizens of the European Union are calling for a global moratorium on the first field release of genetically modified gene drive organisms. The associations Save Our Seeds, the Aurelia Foundation and the Munich Environmental Institute, which are part of the European Stop Gene Drive campaign, handed over a petition to this effect to the German environmental minister Steffi Lemke in Berlin on 31st of May 2022. Enabled by the novel genetic engineering method called gene drive, wild species could be manipulated or even completely eradicated in the future – with unforeseeable consequences for ecosystems.
Gene drives are produced with the help of the new genetic engineering techique CRISPR-Cas. Gene drives can genetically modify or even eradicate entire populations of animals and plants in nature. The so-called gene drive overrides basic principles of evolution and forces the inheritance of a genetic trait to all offspring. This triggers a genetic chain reaction that only stops when all individuals of the affected animal or plant species carry this genetic modification – or have been exterminated. This is intended, for example, to combat disease-carrying insects, invasive species or so-called crop pests in industrial agriculture.
So far, gene drives have only been tested in the laboratory. Now, the research consortium ‘Target Malaria’ in the West African country of Burkina Faso wants to release gene drives into nature for the first time. The goal is to eradicate a mosquitoe species that transmits malaria. But what sounds promising carries enormous risks: once released into the wild, gene drives can neither be retrieved nor can their further development and spread be controlled. If gene drive organisms spread, they could further accelerate the already rapid extinction of species.
At the handover event with German environmental minister Lemke on Leipziger Platz in Berlin, an installation of giant toppling dominoes vividly depicted the risks posed by the gene drive process.
“A genetic chain reaction triggered by Gene Drive organisms could destabilize entire ecosystems and, in extreme cases, cause them to collapse. Every gene drive release – even if it is “only” for experimental purposes – can have unforeseeable and irreversible consequences for pollinator and food webs, already weakened by climate change and high death rate of insects. We urgently need a global gene drive moratorium!”
warns Bernd Rodekohr, manager of the project “Protect the bee from genetic engineering” at the Aurelia Foundation.
“Gene drive organisms do not respect borders and can spread globally,” says the coordinator of the Stop Gene Drive campaign, Mareike Imken. “So far, the global community has neither sufficient knowledge nor binding international agreements under which such a fundamental, irreversible intervention in nature could be regulated.”
The possible use of gene drives is on the agenda of the 15th United Nations Conference of the Parties to the Convention on Biological Diversity (UN CBD), scheduled for autumn in China. EU environment ministers will adopt their common position on the issue in June.
Sophia Guttenberger of the Munich Environmental Institute demands: “Instead of playing Russian roulette with evolution by genetically modifying wild species, we must finally stop the already rapid extinction of species by strengthening the resilience of our ecosystems and stop destroying them everywhere on earth.”
The German envionmental minister Steffi Lemke said at the petition handover:
“I believe that humanity and also science would overestimate themselves with Gene Drives. That’s why I will of course try to reach a position at the Environmental Council of Ministers in June that is based on the European precautionary principle.”
Background:
Gene drive technology uses genetic engineering methods such as the ‘gene scissors’ CRISPR/Cas to introduce certain traits into wild animal and plant populations. For example, if genes that influence fertility or sex are manipulated, entire populations can be wiped out. However, gene drives could also make so-called agricultural pests susceptible to chemical or biological substances or change other characteristics. To do this, both the new trait and the genetic engineering mechanism (CRISPR/Cas) are passed on. In this way, the genetic manipulation continues independently in nature. This “genetic chain reaction” causes all offspring to inherit the desired trait until the entire population or species is genetically modified or eradicated.
Since 2018, the regulation of gene drives has been the subject of controversial debate under the UN Convention on Biological Diversity (UN CBD). At the last Conference of the Parties in Sharm el Sheik, some initial precautionary conditions for release were recommended. But many questions remain unanswered – including, above all, how and by whom the decision on a release of gene drive organisms would have to be taken in view of transboundary spread and unforeseeable ecological, health, economic and social consequences. The existing procedures under the internationally binding Cartagena Protocol of the CBD on Biosafety so far only regulate the intended transfer of genetically modified organisms (e.g. seeds) as products across individual borders. Gene drive organisms, on the other hand, are not products and spread independently in all regions where the target organism is currently present or will be present in the future. In this respect, all potentially affected countries would have to give their consent to a release in advance. Currently, however, only international guidelines for the risk assessment of gene drive organisms and a general process for the technology assessment of new biotechnological processes are on the agenda of the negotiations within the framework of the UN Convention on Biological Diversity. Goal 17 of the planned Global Framework for Biodiversity deals with the prevention of biodiversity damage due to the use of biotechnologies.
Further links:
– For the Stop Gene Drive Campaign’s recommendations on the design of a global gene drive moratorium: https://www.stop-genedrives.eu/en/policy-recommendations/
– To the brochure “Gene Drives. The new dimension of genetic engineering. Applications, risks and regulation.” https://www.stop-genedrives.eu/en/own-publications/
– 15-minute short documentary on the risks and challenges posed by gene drive technology: https://www.youtube.com/watch?v=PLt6ILhQZ7E&t=4s&ab_channel=SaveourSeeds
– All important information about the Stop Gene Drive campaigns on this website and on Twitter.
Press contact:
Mareike Imken
Coordinator of the Stop Gene Drive Campaign
Save our Seeds / Berlin Office of the Future Foundation for Agriculture in the GLS Trust
E-mail: imken[at]saveourseeds.org
Mobile: 0151-53112969
Web: www.stop-genedrives.eu
World Malaria Day 2022
What risks are we willing to take to (maybe) end malaria?
25.04.22 – After a tremendous decrease in malaria cases in the last two decades, malaria is on the rise again, having killed 677.000 people in 2020, among them 80% children under 5. Apart from being deadly, malaria is detrimental for the livelihoods of entire families, communities and countries: farmers not being able to sow their seeds on time, mothers not being able to sell surpluses on markets to earn a living or children not being able to go to school and benefit from education – a vicious cycle of poverty. While this disease affects one third of the world’s population, some scientists suggest that a new technology called gene drive could be a game-changer.
Gene Drives – manipulating the DNA of mosquitoes to pass down an extinction gene
The research consortium Target Malaria, mostly funded by the Bill & Melinda Gates Foundation and the Open Philanthropy Fund, is developing genetically engineered mosquitoes in the lab that would either make all offspring male or all female offspring infertile. They use the Crispr-Cas methodology to implant a system into their DNA that would replicate when mosquitoes mate, ensuring that this gene spreads throughout the wild mosquito population. But while some hope that this would be the magic bullet to suppress mosquitoe populations and stop the malaria transmission cycle, this currently unproven high risk technology poses fundamental questions for humanity: How far are we willing to go, how high can the risks and uncertainties be in order to test a hypothesis?
The Risks of Gene Drive mosquitoes
The risks and consequences of genetic engineering are very hard to predict, especially when the organism is supposed to live and mate in the wild. This is because genes do not only affect the physical shape of animals but also their behavior, their interactions with other species and the way bacteria and parasites affect them. A suppression/eradication gene drive is going to have repercussions along the entire food web and would likely mean that their ecological niche will be occupied by another species, and that the plasmodium parasite (which causes malaria) would lack a host – with unknown consequences. In addition, there is a risk that the modified genes could be passed from the mosquitoes via ‘horizontal gene transfer’ to other species and suppress their population too. If this would affect so called “valued species”, the ecosystems could collapse or be severely damaged.
At this point in research scientists do not know if the genes might be toxic for humans or create allergic reactions. Furthermore, the expected change in behavior of the mosquitoes could lead to increased biting and/or even increased malaria transmission. Also, if humans eat animals that ate the gene drive mosquitoes before they could suffer from secondary toxic effects. Last but not least, if the anopheles gambiae mosquito is eradicated, potentially another mosquito could take its spot, increasing the burden of other diseases.
Same as for humans, gene drive mosquitoes could be toxic for livestock, transmit new diseases or may even – counter-intuitively – increase the transmission of malaria.
Mosquito larvae play an important role in water bodies. Genetically modified larvae could be toxic and negatively impact drinking water and the flora and fauna of water bodies.
As this technology is still very new, naturally studies and discussions on their risks, possible adverse consequences as well as the type of global governance and international regulation needed are only in their infancy. For example, guidance materials for risk assessment have not even been commissioned by the global community. In addition – a plethora of important political, socio-economic, cultural and ethical questions remain unaddressed and unanswered. For example, who should be included in the decision making process and who should be consulted before any release? Would it be enough that a national government such as the Burkinabe government allows such a release and that local village chiefs give their consent? How would decision-making processes be designed to uphold the internationally enshrined rights of Indigenous peoples and local communities to say no to projects that could affect them and their territories? Who would be held responsible and who would have to pay for compensation if the gene drive mosquitoes crossed borders and had adverse impacts on ecosystems or farming in non-target environments?
At the same time there are measures at hand that have in the past been used to end malaria in countries such as, most recently, China and El Salvador, being officially declared malaria free in 2021, preceded by Algeria and Argentina in 2019.
What have been the most successful tools to fight malaria?
Research suggests that the number one tool for the decrease of malaria since 2000 are insecticide-treated bed nets. Approximately 65% of the progress achieved between 2000 and 2015 result from the use of these nets.
Poor Water and Sanitation conditions are associated with a number of diseases, among others with the occurrence of malaria. A better sanitation situation would be a holistic approach to fight malaria, while also fighting diarrhea and respiratory infections that kill millions of children every year. Further research shows that good sanitation and piped water are associated with a lower prevalence of malaria among the population. Dr. Sory shares this opinion and believes that correct drainage systems would heavily lower the burden of malaria.
Artemisinin was rediscovered by the Nobel Prize laureate Tu Youyou who found the cure for malaria in traditional books. It is a component from the Artemisia plant. Drugs against Malaria now often contain artemisinin and can cure all malaria strains present nowadays. Initial research suggests that a tea prepared with the Artemisia plant can have preventive and curative effects. It seems that another plant of the Artemisia family, Artemisia afra, could have similar effects, without containing artemisinin. Lucile Cornet-Vernet and Arnaud Nouvion said that more clinical studies are needed to prove once and for all that these plants work. Until now the WHO asks to not use the plant as tea to not cause resistance to artemisinin. Resistances to artemisinin have been discovered in South-East Asia, but not in Africa so far. Lucile Cornet-Vernet on the other hand points to the fact that in China, this plant has been used for about 2000 years and no resistance has been discovered there yet. Furthermore, the plant comes with a variety of components that could potentially cure malaria, thereby being a “poly-therapy”. Access to health care providers diagnosing malaria and prescribing the drug and the financial means to afford them are the limiting factor here. Or else access to seeds or the Artemisia leaves to cure oneself could be useful, if clinical studies can be conducted and no link to the creation of resistance can be drawn.
There is a multitude of repellents that can protect humans between 3-10 hours from mosquito bites. Seen that most mosquitoes bite in the evening/night, this protection is very helpful when going out late. Many of them have chemical ingredients and some have plant-based ingredients. Amongst those, the German society for tropical medicine, travel medicine and global health recommends only the ones with oil from the lemon eucalyptus and points out that for the other natural repellents too little studies have been conducted. This could be a path worth exploring.
Early detection helps people first to get the needed medicine as soon as possible, suffer the least impact of the disease, and second it helps to decrease the risk of a local outbreak in a community.
A new vaccine was recently approved by the WHO for children under 5 years old. A pilot phase was concluded in Ghana, Malawi and Kenya. Employed prior to high transmission periods of malaria this vaccine seems to have positive affects on immunization. It is recommended that children above 5 months get four doses of the vaccine. Over a 4 years follow-up the efficacy of the vaccine against malaria was 36%.
Why is there still malaria in the world then?
To fight malaria the whole toolbox of measures (as mentioned above) needs to be employed, ranging from prevention through nets and repellents, to access to rapid tests to break the infection chain and access to medicine a few hours after being bitten to cure humans. Adding to this; a holistic approach, including urban planning, education, drainage systems and access to health care is needed to fight malaria – as well as many other diseases that trap people in poverty and create a vicious cycle.
What is the Stop Gene Drives Campaign asking for?
In light of the huge variety of to date unassessed possible environmental, health and socio-economic hazards, the potential for economic and political conflict and a plethora of social, ethical and cultural caveats that the environmental use of gene drive technology would entail , the Stop Gene Drive Campaign demands a global moratorium on the release of gene drive organisms. This means that no gene drive organism should be allowed to be released into the environment – not even for field trials – unless a range of conditions have been fulfilled. Read our policy recommendations here.
It seems to us that malaria prevention funds should be directed at strengthening local health care systems, sanitation and education to turn the fight against malaria into an intersectional approach to fight poverty and neglected diseases in general.
__________
Further readings:
Read more about possible gene drive applications here
Read our FAQ about gene drives here
Read more about gene drive regulation here
Read our interviews with experts on malaria prevention here
_________
References:
Connolly, J. B., Mumford, J. D., Fuchs, S., Turner, G., Beech, C., North, A. R., & Burt, A. (2021). Systematic identification of plausible pathways to potential harm via problem formulation for investigational releases of a population suppression gene drive to control the human malaria vector Anopheles gambiae in West Africa. Malaria Journal 2021 20:1, 20(1), 1–69. https://doi.org/10.1186/S12936-021-03674-6
Czechowski, T., Rinaldi, M. A., Famodimu, M. T., Van Veelen, M., Larson, T. R., Winzer, T., … Graham, I. A. (2019). Flavonoid Versus Artemisinin Anti-malarial Activity in Artemisia annua Whole-Leaf Extracts. Frontiers in Plant Science, 10, 984. https://doi.org/10.3389/FPLS.2019.00984/BIBTEX
ENSSER, VDW, & Critical Scientists Switzerland (CSS). (2019). Gene Drives. A report on their science, applications, social aspects, ethics and regulations. Retrieved from https://ensser.org/publications/2019-publications/gene-drives-a-report-on-their-science-applications-social-aspects-ethics-and-regulations/
Guidance on the environmental risk assessment of genetically modified animals. (2013). EFSA Journal, 11(5). https://doi.org/10.2903/J.EFSA.2013.3200
Landier, J., Parker, D. M., Thu, A. M., Carrara, V. I., Lwin, K. M., Bonnington, C. A., … Nosten, F. H. (2016). The role of early detection and treatment in malaria elimination. Malaria Journal, 15(1), 1–8. https://doi.org/10.1186/S12936-016-1399-Y/TABLES/1
Laurens, M. B. (2020). RTS,S/AS01 vaccine (MosquirixTM): an overview. Human Vaccines & Immunotherapeutics, 16(3), 480. https://doi.org/10.1080/21645515.2019.1669415
Malariaprophylaxe und Empfehlungen des Ständigen Ausschusses Reisemedizin (StAR) der DTG. (2021, August). Retrieved April 18, 2022, from https://www.dtg.org/images/Startseite-Download-Box/2021_DTG_Empfehlungen_Malaria.pdf
Maskin, E., Monga, C., Thuilliez, J., & Berthélemy, J. C. (2019). The economics of malaria control in an age of declining aid. Nature Communications 2019 10:1, 10(1), 1–5. https://doi.org/10.1038/s41467-019-09991-4
Okumu, F. O., Govella, N. J., Moore, S. J., Chitnis, N., & Killeen, G. F. (2010). Potential Benefits, Limitations and Target Product-Profiles of Odor-Baited Mosquito Traps for Malaria Control in Africa. PLOS ONE, 5(7), e11573. https://doi.org/10.1371/JOURNAL.PONE.0011573
Prüss-Ustün, A., Wolf, J., Bartram, J., Clasen, T., Cumming, O., Freeman, M. C., … Johnston, R. (2019). Burden of disease from inadequate water, sanitation and hygiene for selected adverse health outcomes: An updated analysis with a focus on low- and middle-income countries. International Journal of Hygiene and Environmental Health, 222(5), 765–777. https://doi.org/10.1016/J.IJHEH.2019.05.004
Q&A on RTS,S malaria vaccine. (n.d.). Retrieved April 18, 2022, from https://www.who.int/news-room/questions-and-answers/item/q-a-on-rts-s-malaria-vaccine
Target Malaria. (n.d.). The Science: What is gene drive? Retrieved from www.targetmalaria.org/ourwork
Target Malaria | Together we can end malaria. (n.d.). Retrieved April 25, 2022, from https://targetmalaria.org/
The Nobel Prize | Women who changed science | Tu Youyou. (n.d.). Retrieved April 19, 2022, from https://www.nobelprize.org/womenwhochangedscience/stories/tu-youyou
WHO. (2019). The use of non-pharmaceutical forms of Artemisia. Retrieved April 19, 2022, from https://www.who.int/news/item/10-10-2019-the-use-of-non-pharmaceutical-forms-of-artemisia
WHO. (2022). Countries and territories certified malaria-free by WHO. Retrieved April 18, 2022, from https://www.who.int/teams/global-malaria-programme/elimination/countries-and-territories-certified-malaria-free-by-who?msclkid=949a737cbf0711ec9474c0ab673942f3
Yang, D., He, Y., Wu, B., Deng, Y., Li, M., Yang, Q., … Liu, Y. (2020). Drinking water and sanitation conditions are associated with the risk of malaria among children under five years old in sub-Saharan Africa: A logistic regression model analysis of national survey data. Journal of Advanced Research, 21, 1–13. https://doi.org/10.1016/J.JARE.2019.09.001
Yasri, S., & Wiwanitkit, V. (2021). Artemisinin resistance: an important emerging clinical problem in tropical medicine. International Journal of Physiology, Pathophysiology and Pharmacology, 13(6), 152. Retrieved from /pmc/articles/PMC8784654/
International negotiations on Gene Drives resume in person
15.04.22, Berlin -The UN Convention on Biological Diversity (UN CBD) and its sub-protocols are the world's most important forums for establishing internationally binding regulations for Gene Drive technology. For the first time since the beginning of the COVID pandemic - after more than two years of repeated postponements and online meetings - government officials, civil society, scientists and business lobbyists met in person to resume international negotiations in Geneva (Switzerland) between 14-29 March 2022.
During the two and a half conference weeks three different committees discussed a broad range of issues that had previously only been discussed in several online meetings.
At the centre of the Geneva meetings were negotiations of the so-called Post 2020 Global Biodiversity Framework (GBF) to stop and reverse global biodiversity loss through a shared set of goals and measures by 2050. This agreement will be finalized and voted on at the 15th meeting of the Parties (COP 15) of the UN CBD now scheduled to be held in Kunming, China, in August 2022.
Global Biodiversity Framework, Target 17 - Preventing harm from biotechnologies
With regard to the regulation of biotechnologies such as Gene Drives, discussions around Target 17 were of particular relevance. This target is meant to strengthen measures to protect biodiversity from risks and negative impacts emerging from the use of biotechnologies. The Stop Gene Drive Campaign, as part of a group of like-minded civil society organizations named the CBD Alliance, called for a process to anticipate future technological developments, monitor emerging (bio-)technologies and enable the regulation of these biotechnologies to prevent any harmful impacts. The group also stressed the need to uphold the rights of potentially affected indigenous peoples and local communities - especially the right to say no to the use of biotechnologies that could negatively affect their lands, territories and waters. These rules should also stipulate how damage that nonetheless occurs should be compensated for.
While parties such as Bolivia, Ethiopia and Mexico asked for these elements to be included in the text, other parties, most prominently Brazil, tried to undermine the purpose of this target by including text on the potential benefits that biotechnologies could have for biodiversity. While the CBD Alliance called to monitor a broad range of technologies under this target - some parties wanted to narrow down the types of technologies to be covered by including very specific definitions. This is the report of co-leads of contact group 4 that reflects among other these diverse inputs to the discussion.
SBSTTA - Agenda items 4 and 5: Assessing Gene Drive technology
While discussions around the GBF were mostly held by a committee called the ‘Open Ended Working Group’ Geneva also hosted the meeting of an advisory body to the CBD called ‘Subsidiary Body on Scientific, Technical Technological Advice’ (SBSTTA) that discusses general and long-standing issues under the Convention on Biological Diversity and prepares texts to be adopted by the COP. Some of them are of particular interest with regard to regulating Gene Drive technology:
- Agenda item 4 deals with the topic of synthetic biology, which is an emerging field of biotechnology that seeks to redesign or create new living organisms not existing in nature.
The current state of negotiations on this agenda item are reflected in the draft recommendations on synthetic biology and are the results of online negotiations held in April and May 2021. Discussions focused on the establishment of a process under the CBD to anticipate (i.e. ‘horizon scan’), monitor and assess new technological developments in the field of synthetic biology (such as Gene Drives) and their potential impacts for the protection of biodiversity. The Stop Gene Drive Campaign welcomes the establishment of such a long-term process and encourages the formation of a multidisciplinary technical expert group (MTEG), including transdisciplinary experts that represent a broad range of knowledge systems, in order to assess the potential impacts of these technologies. The Stop Gene Drive Campaign stresses that the assessment process needs to take into account socio-economic, cultural, ethical and health questions. The Stop Gene Drive Campaign also demands to reaffirm the need for a highly precautionary approach regarding the release of Gene Drive organisms into nature and to establish further conditions to be met before any environmental release should even be considered. Due to time constraints this agenda item has not been discussed in Geneva but passed over for further discussion at COP.
- Agenda item 5 deals with the risk assessment of genetically engineered organisms (referred to as living modified organisms, (LMOs)). This particular field is subject to a legally binding sub-protocol of the Convention called the ‘Cartagena Protocol’ signed by most but not all of the parties to the Convention.
The current state of negotiations in the draft recommendations on risk assessment is the result of virtual online negotiations held in April and May 2021. Discussions focused on the question whether (additional) voluntary guidance materials on the environmental risk assessment of Gene Drive organisms should be drawn up. Contentious issues were the scope of these guidance materials and the composition of the drafting group. The Stop Gene Drive Campaign welcomes the establishment of such guidance materials. They should address the specific risks of Gene Drive Organisms in general (as opposed to guidance covering only Gene Drive Mosquitoes). The recommendations should be drafted by a diverse and transdisciplinary group of experts, including civil society and indigenous peoples organizations and operationalize the precautionary principle. Due to time constraints this agenda item has not been discussed in Geneva but passed over for further discussion at COP.
- Agenda Item 6 deals with the topic of invasive alien species, which are considered one of the three major reasons for biodiversity loss. In this context Gene Drives have been proposed by some as a technology to combat invasive alien species.
During discussions on this agenda item in Geneva, Gene Drives have been included in the draft recommendations on invasive species which now demands that when considering Gene Drives to fight invasive species, the precautionary approach should be applied. Further discussions on this will be held at COP.
Next steps
The agenda in Geneva was very dense and parties did not manage to finalize their discussions on most of the texts for the GBF. Therefore the CBD Secretariat has announced to hold further meetings. The ‘Open Ended Working Group’ (OEWG) will therefore continue to discuss the targets (such as target 17) of the GBF from the 21st to the 26th of June in Nairobi, Kenya. Another meeting will be held in Bonn from 29 June to 1 July 2022 for the so called ‘Subsidiary Body on Implementation’ (SBI) to discuss indicators to monitor if the new GBF targets are being properly implemented.
Progress in both meetings will be key so that the framework can be accepted by all parties at the COP to be held in Kunming around August and guarantee a robust agenda to halt and reverse biodiversity loss for the upcoming years.
Links and Resources
- Draft recommendation on synthetic biology (CBD/SBSTTA/24/L.5)
- Draft recommendation on risk assessment (CBD/SBSTTA/24/L.6)
- Draft recommendation on invasive alien species (CBD/SBSTTA/24/L.8)
- Co-leads’ proposals on target 17 of the global biodiversity framework: tools and solutions for implementation and mainstreaming, contact group 4; Non-paper of 23-03-2022; Report by the co-leads of contact group 4, including consolidated text on Target 17, reflecting discussions until 2.09.2021 (CBD/WG2020/3/CG/4/REPORT)
World Wildlife Day
World Wildlife Day:
Dreams & nightmares of genetically engineering wildlife.
Why conservation organisations across the world need to speak up!
Most people in the EU – including civil society organisations – are opposed to genetically manipulating food crops but unaware that the scope of genetic engineering projects has shifted radically in the past decade. With the advent of CRISPR/Cas genetic engineering has been brought to a new level while the previously used ‘gene guns’ that enabled for example Monsantos pesticide resistant corn have become quite outdated. With CRISPR many more species – and not only domesticated ones – can be genetically modified in much more targeted and profound way.
Genetic engineering in conservation?
Impressed by these new possibilities molecular biologists and even some conservation organisations have started to dream of genetic engineering as the magic bullet for nature conservation. Invasive species in particular are subject to research projects which aim to develop so called gene drive organisms. Gene drives – a specific application of CRISPR/Cas-based genetic engineering – ensures that a genetically engineered trait will be inherited by 100% of all offspring of an organism. Gene drive organisms, once released into natural environments, are designed to mate with their wild relatives and make the genetic modification a prevalent trait in the wild population – across generations.
One of the main proponents for using gene drives for invasive species elimination is the conservation organisation called Island Conservation. They have a long record of removing non-native invasive predators – predominantly rodents that threaten birds – from tropical biodiverse islands such as Hawaii and Galapagos. To date, this has been done using conventional methods, but Island Conservation believes that other tools such as gene drives are required. For this reason, Island Conservation initiated the Genetic Biocontrol of Invasive Rodents (GBIRd) project, which is supported by seven universities and non-governmental organizations from the USA and Australia, to investigate the gene drive approach and associated questions.
Mice, squirrels, ferrets, wasps, fruit flies and toads are among the species on the wish to be removed from the ecosystems they invaded and harm. Gene drive developers want to simply add a gene drive to specific genes in the germ cells of these organisms that for example code for the sex of the offspring. This could have the effect that only male or female offspring would be born and the population of the locally undesired species would crash over the course of a few generations.
The first steps to develop a gene drive in mice was taken in 2019 at the University of California in San Diego, USA. This research showed, however, that CRISPR gene drives do not yet work well in mammals.
Have we learned any lessons? Does it make sense to fight invasive species with invasive GMO?
In Queensland, Australia, sugarcane farmers in the past had huge issues with beetles that would destroy their crops. In 1935 cane toads (originally from South America) were introduced to fight the beetles. The cane toads succeeded in suppressing the beetle population but turned into invasive species themselves. Now those toads are a plague and spread throughout Australia as they can poison their predators. Australia’s national scientific research agency (CSIRO) is leading research projects on the elimination of cane toads via gene drive. But who can be sure that with gene drive cane toads history is not going to repeat itself?
A similar approach is pursued by the Roslin Institute in the United Kingdom, where the invasive grey squirrel (imported from North America 150 years ago) has pushed back the native red squirrel and destroys trees and bird nests. The idea here is similar to the one in Australia: A gene drive could either render the offspring infertile or only offspring with one sex would be born.
As Dave Goulson, a professor of biology at Sussex University points out anecdotally:
“We used to eat and persecute red squirrels as pests. We introduced grey squirrels because we thought they were cute. Then they spread and the reds started to decline, so we reversed our opinion, deciding that the reds were now cute and the greys should be killed.”
So would it be a good idea to eliminate the grey squirrel with a gene drive? Here’s just one other idea: Researchers have found that reintroducing almost extinct native predators, such as the pine marten into the UK, would lead to a decline of the grey squirrel and a rise of the red squirrel.
Early warnings: gene drives not suitable for conservation purposes
When New Zealand initially considered to include gene drives as part of their Preditor Free Program to rid the island of invasive species, two gene drive developers in 2017 published an article warning against such a decision.
They warned, that once released, the gene drive organisms, for example mice, could remain on the island for several years. Seen that only a few of these gene drive mice would be needed to infect a whole population, their long existence on the island could enable them to “hitch a ride” to other places.
“If we have learned anything from the spread of invasive species, it is that ecosystems are connected in myriad ways and that a handful of organisms introduced in 1 country may have ramifications well beyond its own borders.”
They also warned that, even if these gene drive mice would not manage to leave the country by travelling along via tradeships or planes, experiences in the field of biocontrol seem to suggest that changes are high that they could be moved deliberately to countries where mice pose high damage to certain industries. For example, in the US alone the total cost of annual losses to rats amounts to US19 billion.
The two authors add, that as gene drive organisms are invasive by design, a handful of escaping rats from islands such as New Zealand to the main land would suffice to eliminate all rat populations, thereby severely damaging ecosystems and biodiversity worldwide. In addition, according to these authors, even developing gene drive organisms in labs within an area where the target species lives is dangerous, as any escape would be fatal.
The way forward: A bigger debate & a global moratorium is needed!
In view of the possibility of using gene drives to remove introduced invasive species from sensitive ecosystems, the International Union for Conservation of Nature (IUCN), has also been discussing this technology since late 2015.
In its Members Assembly at its World Conservation Congress in Marseille in September 2021, the IUCN adopted Resolution 075 that mandates the IUCN to undertake an inclusive and participatory member-driven process to explore the role of genetic engineering and synthetic biology in relation to nature conservation. Based on this exploration, Resolution 075 asks the IUCN to develop a policy on this topic until its next World Congress in 2025.
This process will be an important opportunity for the global conservation scene to learn about these new developments. This process will hopefully provide a space to understand that there are many unanswered questions, knowledge gaps, risks and unassessed ecological aspects, conceptual and legal challenges as well as wider questions such as socio-economic, cultural, ethical and legal impacts associated with the genetic engineering of wildlife that need to be addressed before the IUCN can take a position. This position will send an important message to the ongoing regulatory discussions on the level of the UN CBD.
In the meantime the Stop Gene Drive Campaign is demanding national goverments across the world to impose a global moratorium on the environmental release of (including field trials with) gene drive organisms – as long as these open questions have not been answered and a global consensus on the use of this technology has not be reached.
Read here more about how gene drives work, their risks, about the IUCN discussions, the current state of gene drive regulation and our policy recommendations.
European Parliament: no promotion of genetic technologies in development policy!
On Oct. 6, 2021, the European Parliament, in its plenary session, called on the EU Commission and EU member states, through its report on "The role of development policy in combating biodiversity loss in developing countries in the context of the implementation of the 2030 Agenda, "to actively protect the rights of future generations, not to promote genetic technologies with development aid funds and, in particular, not to allow the release of Gene Drive organisms.
Mareike Imken, coordinator of the European Stop Gene Drive campaign, welcomes this resolution:
"Here, for the third time in a row, the European Parliament reinforces its demand not to use Gene Drive technology for precautionary reasons. This demand is also important because the first field trials with the Gene Drive technology are to be implemented in the next few years in Burkina Faso by the Target Malaria project consortium." As noble as the goal thus pursued is to fight malaria - it is also important not to take lightly the unpredictable and potentially catastrophic consequences of cross-border, uncontrollable and irreversible genetic modification or eradication of mosquitoes. I urge the EU Commission and the EU Member States to implement the demands of the European Parliament nationally and internationally!" said Imken.
In paragraph 32, the European Parliament expands on its demand of June 8, 2021, from the EU Biodiversity Strategy and its resolution of January 16, 2020, on the 15th Conference of the Parties to the UN Convention on Biological Diversity:
"[The European Parliament] determines that gene drive technology, such as in genetically modified mosquitoes to control vector-borne diseases, constitutes serious and emerging threats to the environment and nature, including irreversible changes in food supply chains and ecosystems, and losses of biodiversity - a diversity on which the world's poorest depend for their livelihoods. Reiterates its concern about the new legal, environmental, biosafety, and governance challenges that could result from the release of organisms modified by Gene Drive into the environment, even if the release is for conservation purposes; Reaffirms that the free, prior and informed consent of indigenous peoples and local communities must be obtained before introducing technologies that may affect their traditional knowledge, innovation, habits and livelihoods, as well as land use and resource and water consumption; insists that in doing so, all populations potentially affected must be involved in advance in a participatory manner; Considers that gene drive technologies raise concerns about the difficulties of predicting the behavior of affected organisms and that gene drive modified organisms could themselves become invasive species, and therefore, in accordance with the precautionary principle, the release of gene drive modified organisms should not be permitted, even for the purpose of conservation of nature."
From Mareike Imken's point of view, it would be an important further step, also in view of the bad experiences with patented genetically modified seeds in Africa and Latin America, to implement the demand in paragraph 28 of the European Parliament in national development aid programs. In paragraph 28, the European Parliament urges the Commission and Member States to "take into account the Union's obligations under international conventions and also to ensure that no genetic modification technologies are promoted in developing countries with development aid funds."
This resolution is a non-binding opinion of the European Parliament with recommendations to the EU Commission and EU Member States for their international cooperation and work in international conventions such as UN CBD, UNEP, FAO and trade agreements. To implement these recommendations, the EU Commission would have to take them up in its own legislative proposal, which would then have to be confirmed by the European Parliament and EU Member States. However, these recommendations could also find their way into less formally agreed negotiating positions of the EU in its international work.
--------------------------
On the resolution:
- Resolution: European Parliament on the role of development policy in addressing biodiversity loss in developing countries in the context of the implementation of the 2030 Agenda (2020/2274(INI)) - Para. 32 on Gene Drives. (06.10.2021)
- Resolution: Previous European Parliament resolution on Gene Drives: Report on "EU Biodiversity Strategy for 2030: More space for nature in our lives (2020/2273(INI)) - Para 148 on Gene Drives.
- Resolution: European Parliament resolution on the 15th meeting of the Conference of the Parties (COP15) to the Convention on Biological Diversity (2019/2824(RSP)) - para. 15 on Gene Drives (16.01.2021)
European Parliament calls for ban on gene drive technology
Report on the EU' Biodiversity Strategy for 2030: Precaution prevails
9.06.2021, Berlin -The European Parliament yesterday confirmed it‘s precautionary stance towards the use of a new genetic engineering technology called gene drive.[i] In it´s report on the EU’s Biodiversity Strategy for 2030, adopted at the European Parliament’s plenary on 08.06.2021, Parliamentarians demand that „no releases of genetically engineered gene drive organisms should be allowed, including for nature conservation purposes, in line with the precautionary principle.“
Mareike Imken, coordinator of the European Stop Gene Drive Campaign welcomes this decision and comments:„With its position today, the European Parliament recognizes that this technology raises a series of scientific, regulatory, societal and ethical questions and concerns. As its use could severely harm biodiversity, the European Parliament calls to postpone any environmental releases until these questions have been addressed and settled. This is an important message that should feed into the ongoing discussions about global regulations at the next meeting of the International Union for Conservation of Nature (IUCN) in September in Marseille and those of the Convention on Biological Diversity in October in Kunming, China.“
27civil society and science organizations from across the EU had sent a letter to Parliamentarians in support of the amendment ahead of the vote. It „provides reasonable suggestions on how to implement the European Parliament’s previous position in its resolution on the 15th meeting of the Conference of Parties (COP15) to the Convention on Biological Diversity (2019/2824(RSP)“.
In that previous position, adopted in January 2020, the European Parliament had called “on the Commission and the Member States to call for a global moratorium at the COP15 on releases of gene drive organisms into nature, including field trials, in order to prevent these new technologies from being released prematurely and to uphold the precautionary principle, which is enshrined in the Treaty on the Functioning of the European Union as well as the CBD“.
Background information:
Text adopted in the report on the EU Biodiversity Strategy for 2030: Bringing nature back into our lives (2020/2273(INI)):[ii]
The European Parliament,
148. “Is concerned about the new legal, environmental, biosafety and governance challenges that might arise from the release of genetically engineered gene drive organisms into the environment, including for nature conservation purposes; acknowledges the outcome of the Ad Hoc Technical Expert Group of the Convention on Biological Diversity on gene drives and living modified fish, which raises concerns about the difficulties of predicting their behavior, assessing their risks and controlling them after release; notes that gene drive organisms could become invasive species in themselves; considers that global and EU-level risk assessment guidance materials, tools and an environmental monitoring framework, as well as clear global governance and effective mechanisms for controlling and reversing the effects of gene drive organisms, should be fully developed, and that additional research is required on the health, environmental, ecological, ethical and other implications of gene drive organisms to better understand their potential impact; considers therefore that no releases of genetically engineered gene drive organisms should be allowed, including for nature conservation purposes, in line with the precautionary principle ; “.
---------------
Download the press release here.
Letter to MEPs by civil society organisations prior to the vote.
[i] Voting results and margins for EU Biodiversity Strategy
[ii] Report on the EU Biodiversity Strategy for 2030: Bringing nature back into our lives
World Malaria Day: Do we need gene drives to fight malaria?
Gene drive technology carries high risks. Yet it is being promoted by the Bill & Melinda Gates Foundation as a solution to malaria. On the occasion of World Malaria Day, the Stop Gene Drives campaign is launching a project that presents different perspectives on the issue of malaria control and highlights alternative, possibly less risky approaches and innovations to combat malaria.
Infectious diseases such as malaria, dengue fever and Lyme disease are transmitted to humans by so-called vectors, such as mosquitoes or ticks. In the case of malaria, the pathogen and thus the disease is spread exclusively by Anopheles mosquitoes. A global program of malaria control has so far helped to roll back the disease in many regions of the world. Already 38 countries have been certified malaria-free, but there remain 86 countries where malaria control has not been adequately implemented, resulting in several hundred thousand deaths.
This is where the discussion of Gene Drive technology comes into play. Mosquitoes are genetically modified in the laboratory using a process known as CRISPR/Cas so that they pass on a new trait to all their offspring, even if that trait causes the population or the entire species to become extinct. Gene drive mosquitoes produced in this way are expected to massively reduce the number of Anopheles mosquitoes in Africa and thus prevent the transmission of malaria. Gene drives are therefore being promoted as an effective technological solution to combat malaria.
Mareike Imken, coordinator of the Stop Gene Drive campaign, explains in a short interview what concerns there are about the use of gene drive technology and what the goal of the European Stop Gene Drive campaign is. (To the interview)
A leading role in the development of such gene drives is played by the international research consortium Target Malaria, which is largely funded by the Bill & Melinda Gates Foundation. In an interview with us, Dr. Andreas Wulf of Medico International explains their influence on global health policy and the selection of priorities and measures, including those to combat malaria, that are influenced by them. (To the interview)
Target Malaria's plans to use gene drives have already reached the stage where the first projects have been launched in Africa, including Burkina Faso. In July 2019, Target Malaria conducted its first field tests in Burkina Faso using genetically modified sterile mosquitoes that did not yet carry Gene Drive. These trials were considered a precursor to the release of Gene Drive mosquitoes in a later phase of the project. These and subsequent tests have met with resistance from parts of the population in Burkina Faso, as Ali Tapsoba, human rights activist, explains in his interview with us. (To the interview) He is the spokesperson for civil society resistance to the release of Gene Drives in Burkina Faso. Treatment and profilaxis with Artemisia teas grown in Africa are among the measures he would prefer over the use of Gene Drive technology. However, the World Health Organization (WHO) warns against this. Internationally renowned professor Pamela Weathers of Worcester Polytechnic Institute explains how effective and safe Artemisia tea infusions are for treating or preventing malaria in her interview with us. (To the interview)
Burkina Faso has, according to epidemiologist Dr. Sory, a strong strategy to combat malaria. A cocktail of different measures is needed to fight this disease and decrease other diseases at the same time. Better drainage systems, awareness raising and access to health care are just a few points raised in the interview. (To the interview)
Artemisinin is a component derived from the Artemisia plant and present in most anti-malarial drugs nowadays. Lucile Cornet-Vernet and Arnaud Nouvion from the Maison de l'Artemisia, talk about the potential to use the plant as tea as a preventive and curative method for malaria. Furthermore, they make clear that final clinical studies are needed to get the plant approved by the WHO. (To the interview with Lucile and Arnaud)
162 organizations call for global gene drive moratorium
A broad coalition of 162 organizations has sent an open letter to Frans Timmermans, Vice President of the European Commission. It demands that plants and animals modified using new genetic engineering methods continue to be strictly regulated in the future. Furthermore, the EU Commission should support a global moratorium on gene drive organisms.
The existing EU genetic engineering standards ensure the implementation of the precautionary principle and protect the environment and consumers, wrote the organizations from the fields of environmental and consumer protection, agriculture and the food industry. Farmers and consumers would be free to choose whether to eat or grow genetically modified crops. The current occasion for the open letter is a study on the current status and future regulation of genetically modified organisms in the European Union (EU). The governments of the European member states had asked the EU Commission in November 2019 to prepare such a study. It should take into account various aspects, including scientific progress, the legal situation, as well as a publication of the "European Group on Ethics in Science and New Technologies." The European Commission has announced that they will publish the study at the end of April.
The authors of the letter to Timmermans and other members of the EU Commission also call on the Commission to ensure that the regulation of genetic engineering in the UK remains EU-compliant after Brexit. This is because the British government is currently planning to revise its genetic engineering legislation. As reported by Infodienst, it invited stakeholders to a consultation process at the beginning of the year, which ended in mid-March. The organizations are now calling on Timmermans to lobby the UK government to drop these plans. If genetically modified plants were to be less strictly regulated in the UK in the future, this would also affect the trade with the EU.
Finally, the 162 organizations call for the EU Commission to support a worldwide moratorium on the use of so-called gene drive organisms. Gene drive organisms result from a special application of new genetic engineering. This poses the risk that entire species of organisms could be severely decimated or wiped out. The European Parliament had already spoken out in favor of a global moratorium in January 2020 in the form of a petition. Especially in times of "ecological crisis, when a million species are threatened," experiments with a technology that is also referred to as "extinction on demand" cannot be carried out, according to the justification.
- Press release on letter by IFOAM and others to EU Com Vice-President Timmermans: Don’t deregulate GM crops & animals. (30.03.2021)
- Letter by IFOAM EU and others to EU COM Vice President Timmermans: Don't deregulate GM crops & animals (30.03.2021)
- Answer by European Commission to the letter by IFOAM EU and others (06.05.2021)Lett
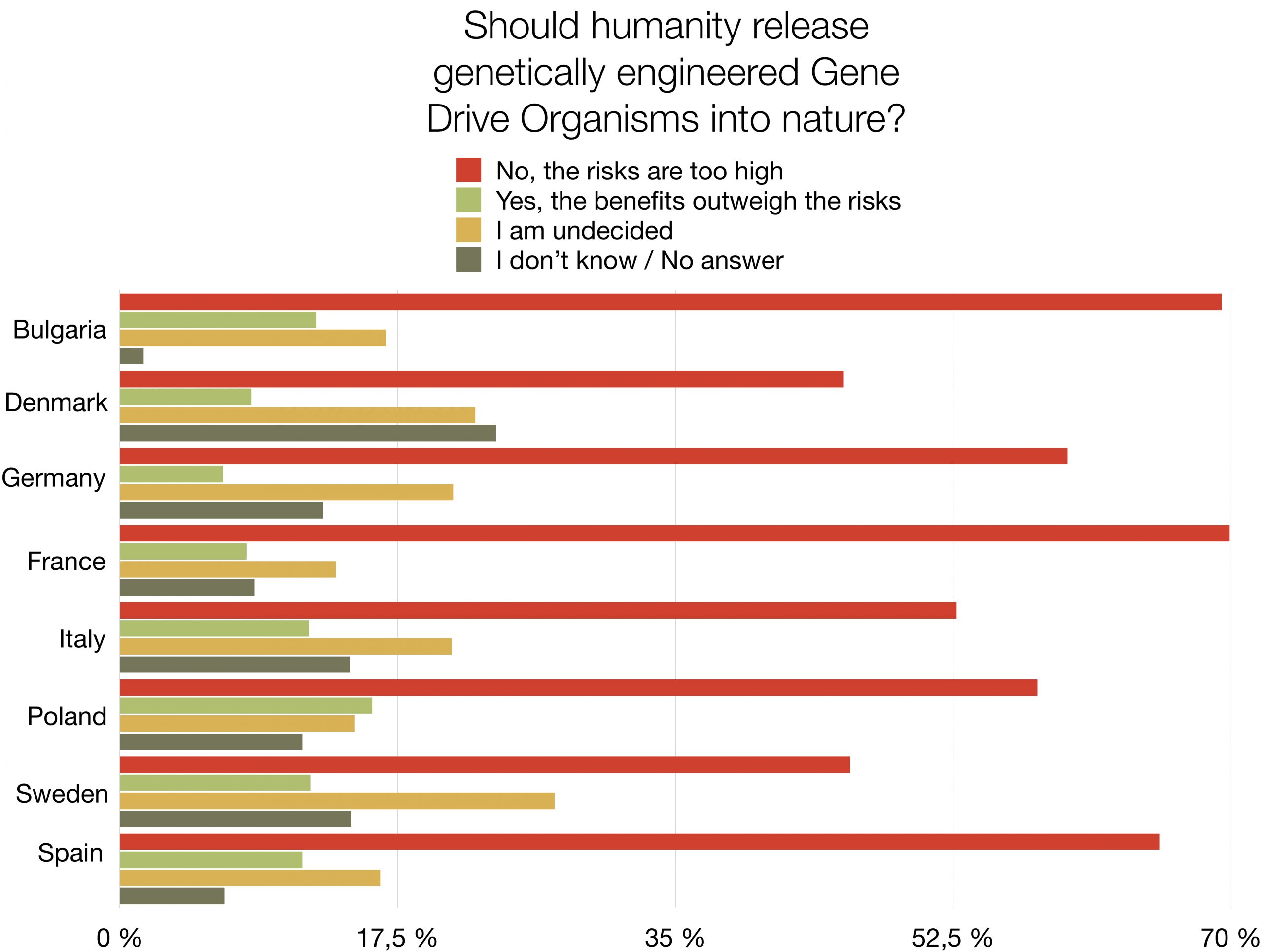
Survey: EU citizens reject genetic engineering of wild species with Gene Drives
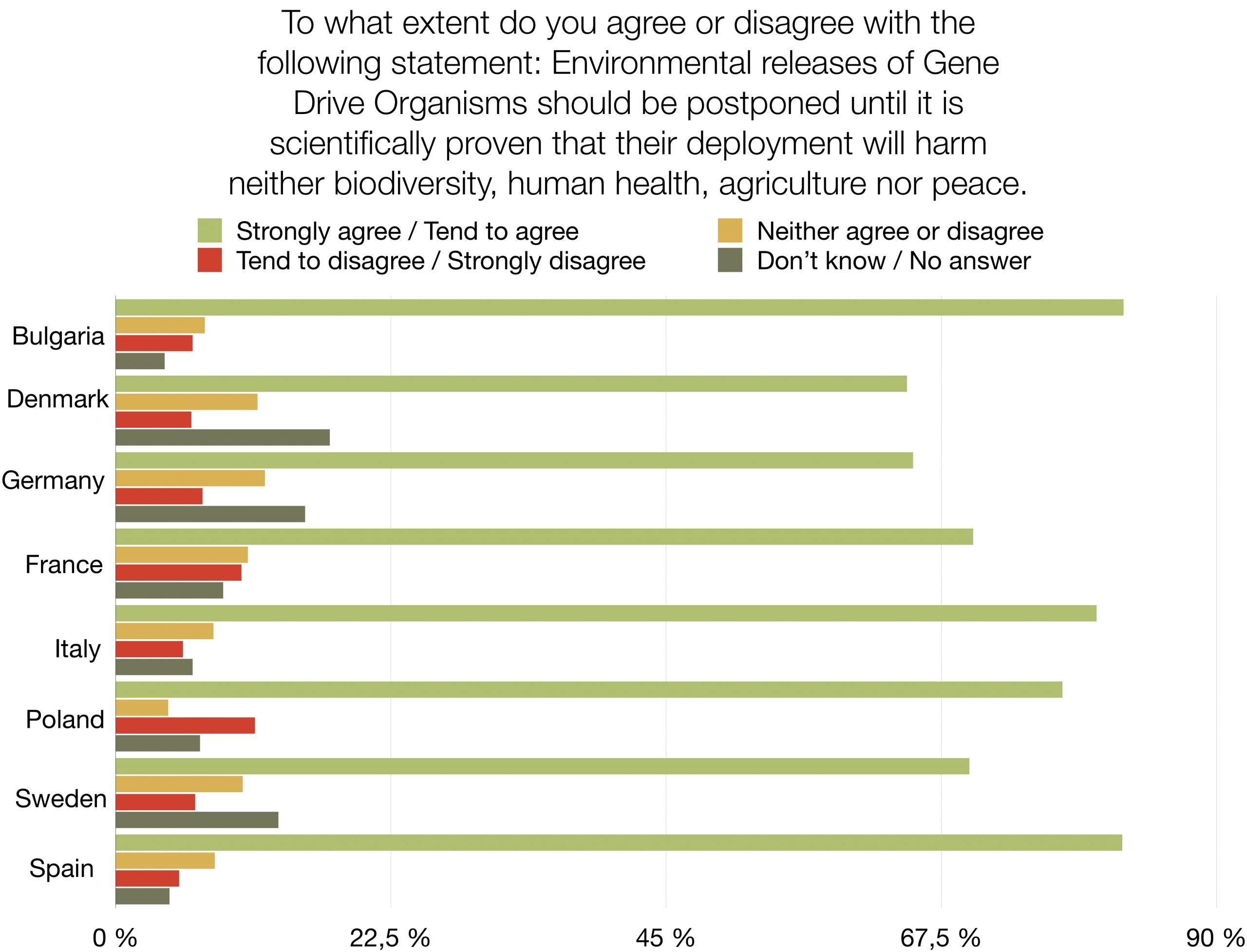
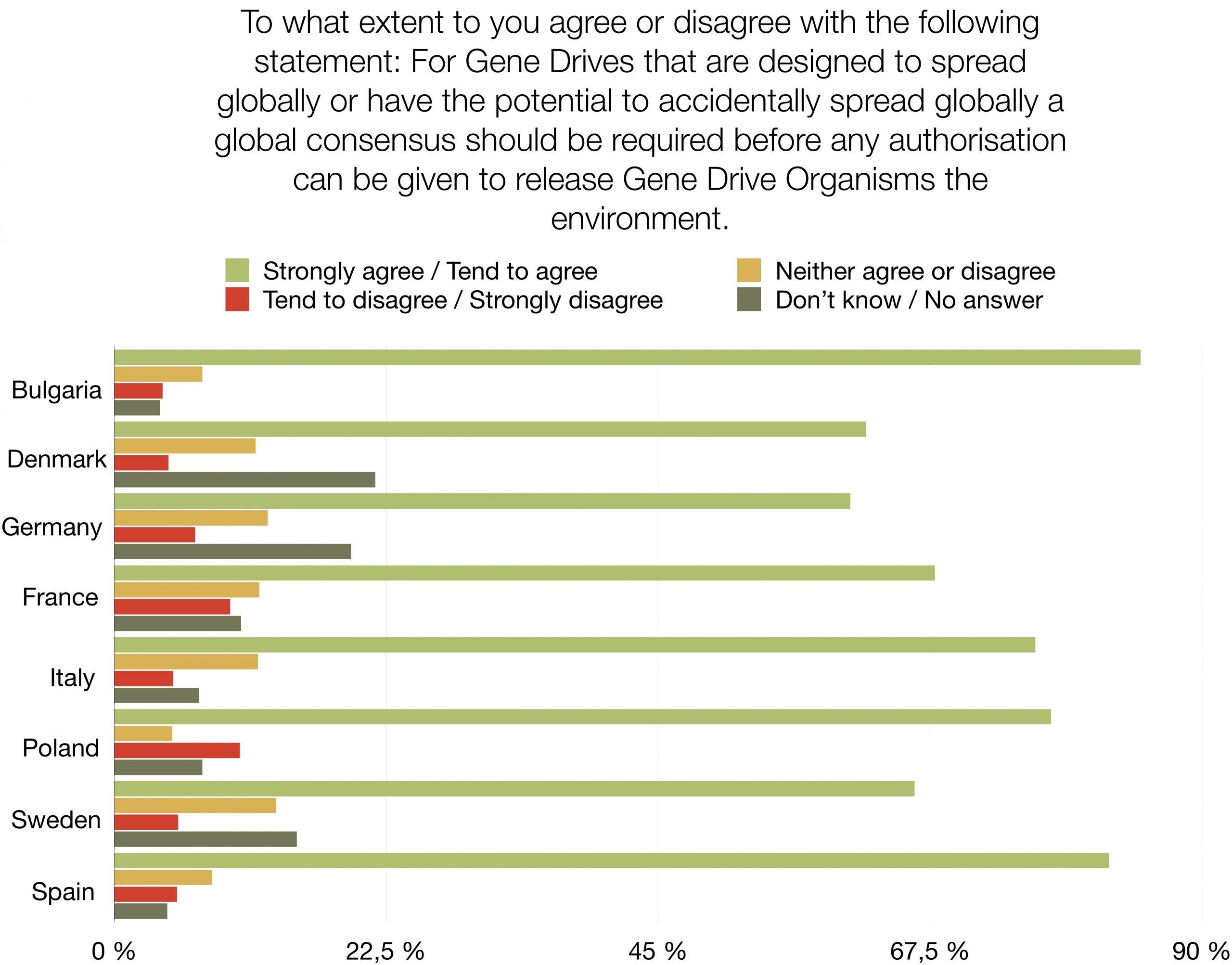
Should humanity release genetically engineered gene drive organisms into nature? The response of a majority of citizens in eight European countries is: “No, the risks are too high”. This first opinion poll on the subject shows high levels of rejection (46% - 70%) and very low levels of support (7% - 16%) for the use of Gene Drive technology in the environment. The survey amongst nearly 9.000 people is representative of 280 million EU citizens. It was commissioned by nine NGOs demanding an informed and inclusive public debate and a global moratorium on the environmental release of this new type of genetically modified organisms. The survey also reveals that a large proportion of respondents is still undecided (14% - 27%) or did not know how to answer (1% - 24%).
"Such a powerful technology with potentially irreversible consequences for wild species and all their ecosystems must be controlled by strict international rules and procedures of decision making. We do not believe that the release of gene drive organisms from the lab into the environment should happen at all. At least, it would require strict international standards of risk and technology assessment and a prior inclusive, democratic decision making based on precaution and the prior informed consent of all peoples and states potentially affected" states Mareike Imken from Save Our Seeds, Germany, who coordinates the European Stop Gene Drive campaign."
A large majority of respondents (65%-82%) agree that environmental releases of gene drive organisms should be postponed until there is scientific proof that their release would not harm biodiversity, human health, agriculture or peace. A similar majority (61% - 85%) agrees that the authorisation of environmental releases of gene drive organisms that could spread globally should require a global consensus.
This representative survey was conducted by the international market research institute YouGov and polled 8.826 citizens, from 8 EU countries, including Germany, Italy, France, Spain, Poland, Denmark, Sweden and Bulgaria in December 2020. It was commissioned by WeMove Europe, Save Our Seeds (Germany), Skiftet (Sweden), France Nature Environnement (FNE) (France), POLLINIS (France), OGM Dangers (France), Bund für Umwelt und Naturschutz (BUND) (Germany), Deutscher Naturschutzring (DNR) (Germany), Umweltinstitut München (Germany), Za Zemiata (Bulgaria).
Downloads
Download YouGov Results Gene-Drive Opinion Poll | PDF
Download Gene Drive Acceptance Survey - A graphic overview of the results | PDF
Further information
EU-wide petition for global gene drive moratorium
Open letter by over 200 organizations worldwide calling for global gene drive moratorium
Resolution of the European Parliament for a global gene drive moratorium
Online-Discussion: Gene Drives - Protecting People and Nature through Genetic Extermination?
Ricarda Steinbrecher, geneticist and board member of the European Network for Scientists for Social and Environmental Responsibility (ENSSER), emphasized that it is difficult or even impossible to make reliable predictions about the effects of a future application of Gene Drives, especially at the current time. After all, organisms are released which then independently carry out the genetic modifications in each generation. "Mistakes can be made every time. Every time, something else can be added." To ensure the preservation of biodiversity, new technologies such as Gene Drives must be looked at very closely to ensure that they do not pose any risks to our ecosystems. This is why she strongly advocated the precautionary principle during the international negotiations on the regulation of gene drive technology at the UN Convention on Biological Diversity. As a long-standing scientific advisor and participant in expert groups within the UN Convention on Biological Diversity, she reported that there is a strong influence of lobby groups on these expert bodies: For example, the Bill & Melinda Gates Foundation, one of the main financiers of the technology, had invested 1.6 million dollars in a PR agency to increase the acceptance of Gene Drives. She concluded: "The pressure to implement this technology is not commensurate with the risk."
Ali Tapsoba de Goamma, human rights and environmental activist from Burkina Faso and spokesperson for a civil society association of 40 organizations for agroecology and against Gene Drives (CCAE), reported that since 2012, the project Target Malaria has been preparing to decimate malaria-transmitting mosquitoes in Burkina Faso by means of Gene Drives. In 2019, the first field tests with genetically modified male mosquitoes that are not capable of reproducing took place, but not any Gene Drive Organisms have been released yet. Ali Tapsoba de Goamma criticized that Target Malaria had obtained the consent of the government and village leaders for these tests, but not the consent of the entire population of Burkina Faso. Rather, they had taken advantage of the fact that there are so many illiterate people in the local villages. The majority of the inhabitants of Burkina Faso are against these experiments. He raised the question: "Why not try this first in scientifically better equipped countries, but in Burkina Faso?" In his view, Burkina Faso was in a position to combat malaria itself. He said that this does not require gene drives, but a good health concept.
Dr. Andreas Wulf, doctor and consultant for global health at the medical emergency aid organization Medico International, emphasized that epidemics like malaria require long-term strategies. The idea of trying to solve such a disease with the one-time use of a technology without continued commitment is questionable, he said. One should not rely on such a "technological fix". Experience in combating other epidemics has shown that the success of the measures depends on good cooperation with the people on the ground and finding local solutions. He also criticized from a democratic point of view: "It is a problem that so much decision-making power is given to these private actors, the companies / foundations. A handful of people choose which area of research to invest in. In addition, the media coverage of research is also financially supported by these foundations". Dr. Andreas Wulf concluded that these private funds need to be embedded in the public health systems.
Mareike Imken, head of the Stop Gene Drive campaign of Save Our Seeds, explained that with the European campaign, which is supported by many organizations throughout Europe, she wanted to initiate a critical discussion in society and politics about "whether and if so, under what circumstances we want to use this technology and what restrictions it needs."
She went on to explain: "Gene Drives provides us as humanity with a tool to specifically eradicate or change wild species. In times of a species extinction that is existential for mankind, this must be considered and ethically discussed. This should not be decided lightly by those few people". She also points out that "the knowledge about Gene Drives is not yet sufficiently advanced".
In order to have time for this discussion, in-depth risk research including technology assessment and the development of internationally valid rules and decision-making mechanisms, a global moratorium on the release of gene drive organisms is needed. This moratorium must be adopted at the next Conference of the Parties of the UN Convention on Biological Diversity (COP 15).